Discover the detailed characteristics and classifications of organometallic compounds, including their physical, chemical properties, and industrial uses
The study of organometallic compounds is conducted under the scope of organic chemistry.

- Definition – The Compounds that contain at least one metal-carbon bond are known as organometallic compounds.
- organometallic compounds ⇒ Carbon–Metal bond compounds
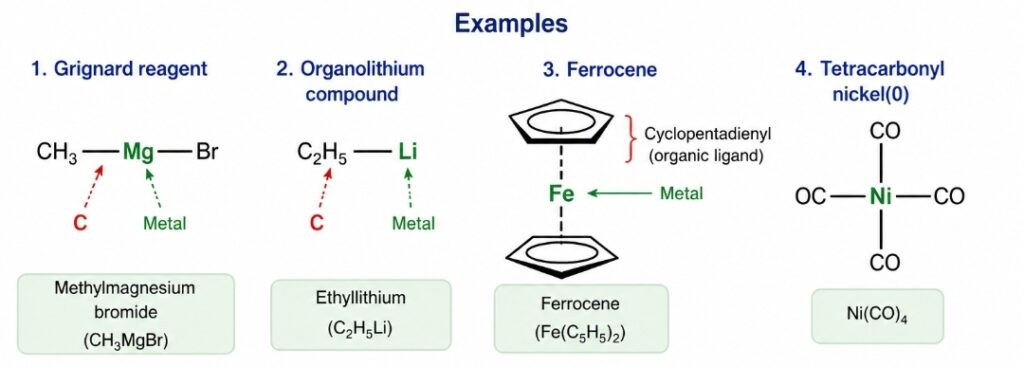
- CH3-Li [Methyl lithium]
- C2H5-MgBr [Grignard reagent]
- (C2H5)4 Pb [Tetraethyl lead]
The bond formed between carbon and a metal can be ionic or covalent.
![]() Example –
Example –
- covalent bond = Pb(C2H5)4
- Ionic bond = [Na+C2H5–]
![]() There are three types of classification of organometallic compounds-
There are three types of classification of organometallic compounds-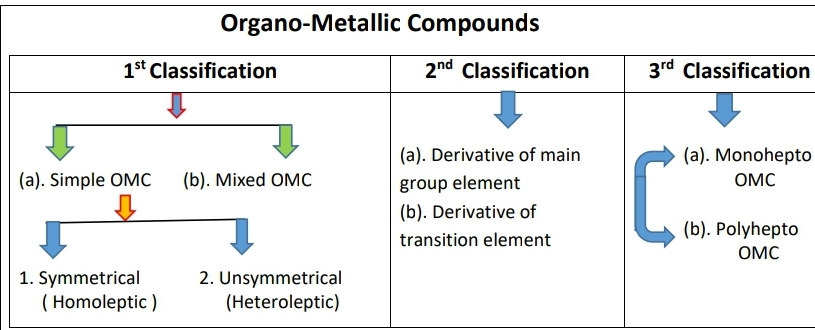
 Classification of Organometallic compounds:–
Classification of Organometallic compounds:–
[A]. 1st classification ⇒
This classification is based on Hydrocarbon + other group. it is divided into two parts-
- Simple OMC – The compounds having only hydrocarbon radicals or hydrocarbon atom attached to the metal atom known as simple organometallic compounds. It is also divided into two parts
- •Homoleptic (symmetrical) – these compounds have similar hydrocarbons.
- example = C2H5-Hg-C2H5 and C2H5Pb-C2H5
- • Heteroleptic (unsymmetrical) – these compounds have different hydrocarbons. example= C2H5-Hg-C4H9
- Mixed OMC – Compounds in which a group other than a hydrocarbon group is also directly bonded to the metal atom are called mixed metallic compounds. example = RMgX
- [B]. 2nd classification ⇒ this classification is on the basis of metal types.
- 1. Derivative of main group element – S Block (except- li, be, mg) P Block (except- s,o,n,p,c,Al)
- 2. Derivative of transition element – d & f block elements forms large no. of λ-derivatives.
[c]. 3rd classification ⇒
this classification is based on the Hapticity. Hapticity can be indicating by the no. of carbon atom in organic moiety. It is also divided into two parts-
1). Monohepto – OMC containing monohepto ligand which linked to metal atom through one carbon only. It is derivatives of main group elements. (s & p blocks)
2). Polyhepto –OMC containing polyhepto ligand which linked to metal atom through more than one carbon atoms. It is derivatives of transition elements. (d & f block)
 Characteristics of Organometallic compounds :-
Characteristics of Organometallic compounds :-
![]() Physical properties –
Physical properties –
- OMC are largely covalent in character as shown by their property.
- OMC mostly exist in the solid states, especially the compounds in which the hydrocarbon groups are aromatic or have a ring structure.
- OMC are found to be to toxic to humans in many cases. specially if the compounds that are volatile in the nature.
- OMC compounds can act as reducing agents.
- OMC are soluble in non-polar or weakly polar organic solvent such as Benzene, Toluene, Ether etc.
 Chemical properties of OrganoMetalic compounds –
Chemical properties of OrganoMetalic compounds –
- 1). Stability – with Temperature (Decomposition). Some are low stable at room temperature, and some are stable at the high temperature.
- example= Me4Ti ( decompose at room temperature)
- Me4Si (stable at 5000 c for many days)
- 2). Oxidation – Organometallic compounds are generally prior to oxidation in air at room temperature.
- example- Si, Ge, Sn, Pb are easily oxidized. [H3C]2MG not oxidized.
- 3) Hydrolysis –
- Group 4 & 5 → not hydrolyzed
- Group 1, 2 & Zn, Cd, Al, Ga →readily hydrolyzed
- 4). Action on non-metallic elements –
RLi + X2 → R-X + LiX
Organometallic compounds play a very important role, these compounds are extensively used in industries, pharmaceuticals and catalysis. organometallic compounds have a direct bond between carbon and the metal which can be covalent or partially ionic. These compounds are mostly useful and very important in many of catalysts process and because of it has a covalent and ionic nature, orbital overlap and hybridization bonding it play a very important role in Nucleophilic reactions and Addition reactions.
![]() Uses and Applications of Organometallic compounds:-
Uses and Applications of Organometallic compounds:-
- Organometallic compounds play an important role in the process of drug synthesis. these are very useful compounds in the pharmaceutical industry.
- OMC also helpful in the process of mechanism of complex molecules.
- presently OMC are also play a very important role in the therapy research of cancer.
- OMC applicable in the mechanism process of daily products like pipes, bottle, chairs etc. and plastics synthesis
- They are used in formation process and the Organic Synthesis for making process of new organic compounds Organometallic compounds very helpful.
- Organometallic compounds are very useful in the mechanism process of the polymers (like-plastic, rubber, pipes etc.) example: – Ziegler-Natta catalysts [ it uses for the polyethylene and polypropylene production]
