what is photodegradation of polymers, learn about types of photodegradation of polymer with definition and mechanism (step-by-step)

Organic polymers are exposed to U.V. or visible radiations in the presence of atmospheric oxygen, Then there organic polymer undergo Photodegradation, which includes a chemical change in the polymer.
It is responsible for the destruction in the mechanical properties of the bulk polymer. Most of the polymers undergo photodegradation which causes chemical, physical or biological changes in the polymer. Photodegradation of polymer has importance from two different aspects-
- Ultraviolet & visible radiations are harmful to biopolymers like DNA, Polysacarides, Proteins etc. an understanding of their mode of photolysis is important in knowing the details of life process.
- More & more use of polymeric material in everyday life has caused a problem of disposal.
• Definition – Photodegradation is the degradation of polymer caused by the absorption of UV light, leading to bond breaking and deterioration of physical and chemical properties.
• Photodegradation process can be photochemically induced, it has two type –
♦ Types of Photodegradations of Polymers ⇒
1. Homolytic Cleavage – The photolysis with homolytic cleavage may cause the elimination reaction at the sight of weaken point of bond.
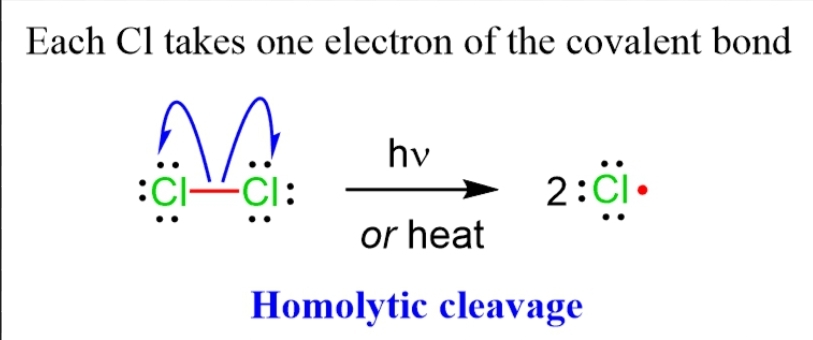
2. Heterolytic Cleavage – The photolysis of this type also occurs at weaken bond & this type of cleavage may cause free radical formation.
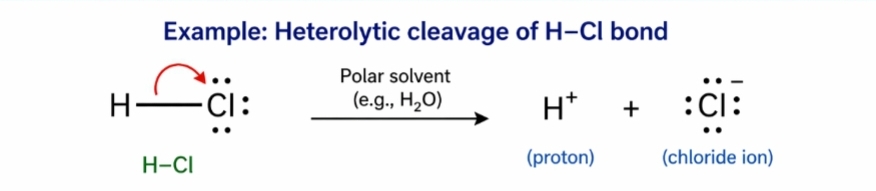
‘The point of bond cleavage may or may not be the seat for light absorption.’
It is important to note that when the molecule dissociates from an excited state, it is called Photolysis.
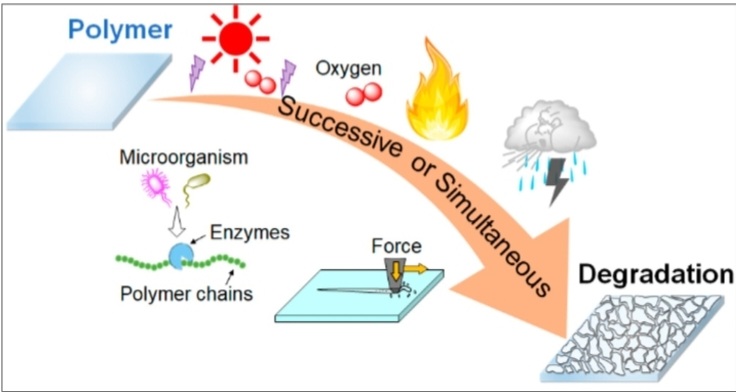
These are most specific types of the photodegradation of polymer –
- Homolytic cleavage – forms free radicals.
- Heterolytic cleavage – forms charged ions.
◊ Mechanism (step-by-step) ⇒
- Initiation- (1) UV light is absorbed by the polymer. (2) Weak bonds break, forming free radicals.
- Propagation – (1) This creates a chain reaction. (2) These radicals attack other polymer chains → more radicals’ form. (3) Free radicals react with oxygen → form peroxy radicals (ROO•)
- Termination – Radicals combine and stop the reaction. However, by this time the polymer structure is already damaged.
 Some common reactions which are involved –
Some common reactions which are involved –
- Chain scission (breaking of polymer backbone)
- Cross – linking (chains joining together)
- Oxidation (reaction with oxygen → carbonyl groups form)
 Effects on Polymers –
Effects on Polymers –
- Loss of mechanical strength
- Brittleness and cracking
- Color fading or yellowing
- Surface chalking
- Reduced lifespan
Example ⇒ Plastic chairs left in sunlight become weak and brittle over time.
![]() The Polymers that are highly affected –
The Polymers that are highly affected –
- Polyethylene (PE)
- Polypropylene (PP)
- Polystyrene (PS)
- Polyvinyl chloride (PVC)
 There are some ideas to prevent photodegradation –
There are some ideas to prevent photodegradation –
- Add UV stabilizers
- Use antioxidants
- Apply protective coatings
- Add pigments like carbon black (absorbs UV)
Photodegradation of polymer is the breakdown of polymer materials when they are exposed to light especially ultraviolet (UV) radiation from sunlight. This process gradually weakens the material, causing loss of strength, discoloration and cracking.
Therefore, Photodegradation is the degradation of polymer caused by the absorption of UV light, leading to bond breaking and deterioration of physical and chemical properties.
