[Learn about what is atoms and molecules? explanation of atoms & molecules with definition, differences & examples.]

- What are atoms and molecules?
Have you ever deeply observed a building wall. the building has many of rooms and every room is a part of building. these walls made a room, and many rooms form a building. did you ever think what is the building block of the huge building? It is a small grain of sand. Similarly, the blocks of all matters are atoms. Molecules are made of the combination of two or more than two atom particles.
♦ Laws of chemical combination – there are two type law of chemical combination-
- Law of conservation of mass
- Law of constant properties
- LAW OF CONSERVATION OF MASS → ”law of conservation of mass states that mass can neither be created nor destroyed in a chemical reaction.” There is no change in mass during a chemical reaction.
- LAW OF CONSTANT PROPORTION→ ”According to this law, In a chemical substance the elements are always present in definite proportion by mass.”
·Definition –
- Atom ⇒ An atom is the smallest unit of any element that forms a chemical element.
- Molecule ⇒ A molecule is a group of two or more atoms joined together by a chemical bond.
♦ ATOM –
- atoms are extremely tiny and very small. atoms are smaller than anything and we can not see with the naked eyes but together they form all matters. entire world is made up of the atoms. examples – • Oxygen atom (o) • Helium atom (He)
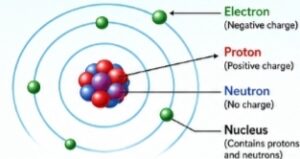
Structure of an Atom- An atom has three main parts which shows its structure –
- Protons – Inside the nucleus (Positive charge)
- Neutrons – Inside the nucleus (No charge)
- Electrons – electrons move around the nucleus (Negative charge)
Atomic radius is measured in nanometers –
- 1/109 nm = 1 nm
- 1 m = 109 nm
Shape and size of atom ⇒ Atoms are extremely very small in size. The size of an atom is indicated by its radius which is called ‘atomic radius’ or (Radius of atom).
- – Atomic radius of some common elements –
| Element | Radius of atom |
| Hydrogen | 0.037 nm |
| Carbon | 0.077 nm |
| Nitrogen | 0.074 nm |
| Oxygen | 0.073 nm |
| Chlorine | 0.099 nm |
| Sulphur | 0.104 nm |
Dalton’s atomic theory –
- All matter is made of very tiny particles called atoms, which participate in chemical reactions.
- Atoms are invisible particles, which can not be created nor destroyed in a chemical reaction.
- Atoms of a given element are identical in mass and chemical properties.
- Atoms of different element have different masses and chemical properties.
- Compounds are formed when elements combine in simple, fixed whole number ratios and the kind of element remains same in the compound.
According to Dalton’s atomic theory, all matter, whether an element or a mixture is composed of small particles called atom.
♦ Molecule –
- Molecule is a group of two or more atoms that are chemically bonded together.
- In a molecule, two or more atoms are held together strongly by some kind of attractive force or chemical bond.
- It is the smallest particle of an element or compound which can exist freely and shows all the properties of that substance.
Types of molecules-
- Elemental molecules – The molecules which are made of the same type of atoms, known as the elemental molecules. example= Oxygen (O2) , Hydrogen(H2)
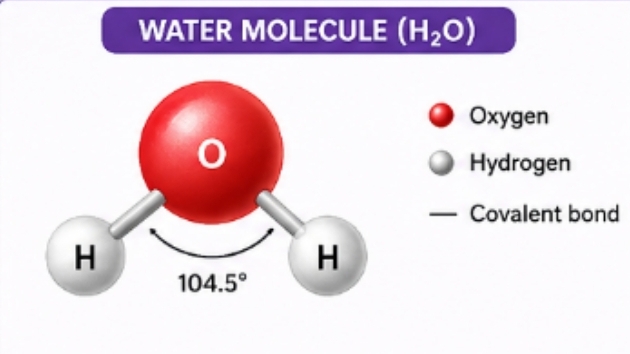
- Compound molecules – The molecules that are made of different types of atoms, called as the compound molecules. example= Water (H2 O)
Molecules of some compounds –
| Compound | Combining Elements | Ratio of Mass |
| Water (H2 O) | Hydrogen, Oxygen | 1 : 8 |
| Ammonia (NH3) | Nitrogen, Hydrogen | 14 : 3 |
| Carbon dioxide (CO2) | Carbon, Oxygen | 3 : 8 |
Importance of Atoms and Molecules –
- Atoms and Molecules are very helpful in understanding all of chemical reactions.
- Atoms and molecules form all the substances in the universe, so it plays an important role towards universe.
- They are useful in the industries and technology.
- It play an important role to easily study about the biology, chemistry and physics.
- Many of the medicines are made by using of the molecules.
♦ Difference between Atom and Molecule –
| Features | Atom | Molecule |
| Definition | Smallest unit of an element | Combination of two or more atoms |
| Existence | may or may not exist freely | exists freely |
| Size | smaller | larger |
| Importance |
Medicine, industries, technology |
Medicine, industries, technology |
| Example | Oxygen (O), Helium (He) | Methane (CH4), Ammonia (NH3) |
Atom and Molecules are the specific foundation of science which helps us to understand the universe. after knowing about the structure of atom and molecules, its easy to understand and calculate the chemical formula of compounds. A chemical formula of compound shows its constituent elements and the number of atoms of each combining element. Air, water, food even human body is made up of tiny particles of atoms and molecules.
